最高のコレクション favipiravir 140213-Favipiravir pi
Favipiravir is a pyrazine carboxamide derivative developed by Toyama Chemical of Japan to act against many RNA viruses It was first described as a selective inhibitor of influenza virus replication with minimal cytotoxicity (Furuta et al, 02)Favipiravir (originally known as T705) is an orally administered novel antiviral compound with a unique mechanism of action that is active against a wide range of RNAbased viruses in laboratory tests Favipiravir has recently being approved in Japan under the brand name Avigan Avigan is an experimental antiviral drug being developed by The Drugs Controller General of India (DCGI) has approved Glenmark Pharmaceuticals, an Indian pharmaceutical company, to sell generic versions of Favipiravir for the treatment of COVID19 This
Favipiravir Very Effective Against Covid 19 Clinical Trials Show Cgtn
Favipiravir pi
Favipiravir pi- Favipiravir may yet turn out to be a drug worth prescribing to COVID19 patients – but we will have to wait for these ongoing trials to finish to know if this is the case As of now, experts say, it's best to hold off The reporting for this article was supported by a grant from the Thakur Family Foundation The foundation did not exercise any editorial control over the contents of the Favipiravir is a broadspectrum antiviral that has shown promise in treatment of influenza virus infections While emergence of resistance has been observed for many antiinfluenza drugs, to date, clinical trials and laboratory studies of favipiravir have not yielded resistant viruses
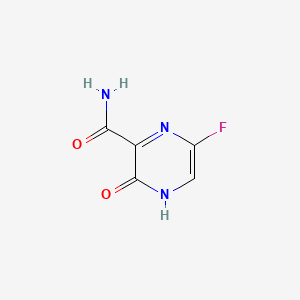



Favipiravir C5h4fn3o2 Pubchem
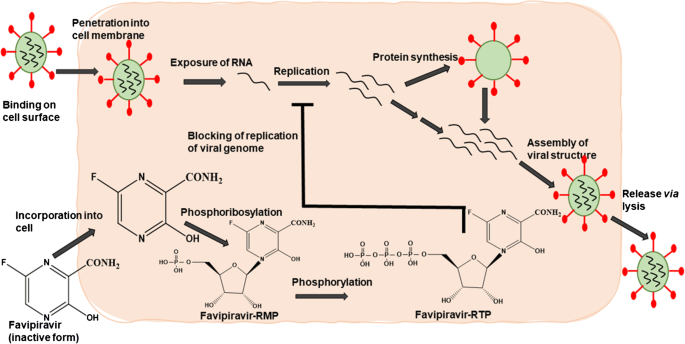
Favipiravir is a pyrazinecarboxamide derivative with activity against RNA viruses Favipiravir is converted to the ribofuranosyltriphosphate derivative by host enzymes and selectively inhibits the influenza viral RNAdependent RNA polymerase6fluoro3hydroxy2pyrazinecarboxamide) is an antiviral agent that selectively and potently inhibits the RNAdependent RNA polymerase (RdRp) of RNA viruses Study arm groups will receive a Favipiravir treatment of 1800 mg PO BID on day 1, then 800 mg PO BID from day 2 onwards and control groups will receive the same quantity of Placebo Treatment will be continued till 5 days after for mild groups and 10 days for moderate groups Eligible patients will be randomly assigned (11) to either Favipiravir or Placebo among mild cases;
Favipiravir is an antiviral drug which has been licenced in Japan since 14 to treat influenza It works by inhibiting a viral enzyme called RNA polymerase, preventing viral replication within human cells This viral enzyme is common to several viruses, including SARSCoV2, which causes COVID19 Favipiravir is quickly emerging as the top choice among 25 drug candidates that the Council of Scientific and Industrial Research (CSIR) is considering for COVID19 treatment, the Ministry of Science and Technology said in a press release yesterday (April 30) Favipiravir is an antiviral drug that was originally made to treat the flu in JapanFavipiravir proves effective in clinical trials As included by the earlier source The Guardian, officials and medical authorities have announced that the drug has been effective in treating
Favipiravir triphosphate is a purine nucleoside analogue, which acts as a competitive inhibitor of RNAdependent RNA polymerase 2 It has activity against influenza A and B, including activity against oseltamivir and zanamivirresistant influenza viruses, several agents of viral haemorrhagic fever and SARSCoV2 in vitro 2–4 Favipiravir is approved for novel epidemicFavipiravir was also trialed for treating Ebola; The antiviral drug Favipiravir (FabiFlu) inhibits a viral enzyme called RNA dependent RNA polymerase (RdRP), thereby stopping the virus' replication cycle
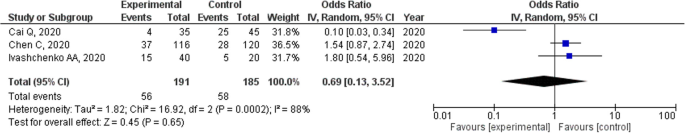



Favipiravir For The Treatment Of Patients With Covid 19 A Systematic Review And Meta Analysis Bmc Infectious Diseases Full Text




Fujifilm Tests Favipiravir As A Covid 19 Treatment
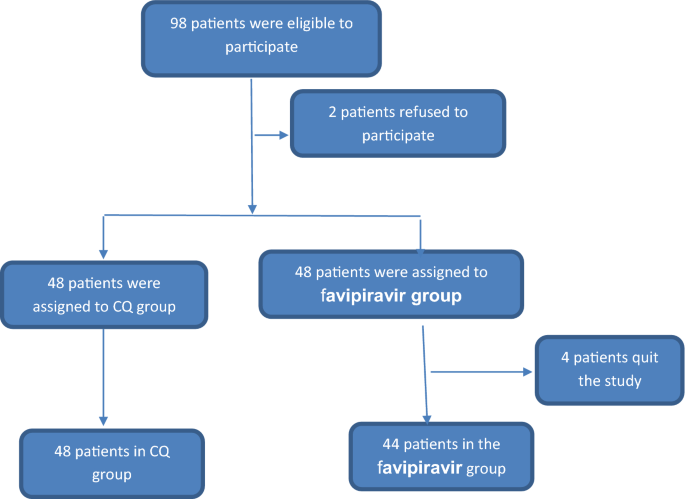
Favipiravir is under investigation for the treatment of coronavirus disease 19 (COVID19) (Cai ; The favipiravir group included 48 patients who received 1600 mg of favipiravir twice a day on the first day and 600 mg twice a day from the second to tenth day, added to the standardofcare therapy for 10 days Four patients in this group quit after the beginning of the study, and the final number in this group was 44 patients The four patients who left the study preferred toFavipiravir definition is an antiviral drug C5H4FN3O2 that is administered orally or intravenously and inhibits viral replication of RNA viruses (such as the virus causing influenza) by interfering with viral RNA polymerase function How to use favipiravir in a sentence




Favipiravir Pharmacokinetics In Nonhuman Primates And Insights For Future Efficacy Studies Of Hemorrhagic Fever Viruses Antimicrobial Agents And Chemotherapy




The Dhaka Trial Clear Cut Evidence Favipiravir Effective Against Covid 19 With Compelling Results
AVIGAN® / REEQONUS™ (favipiravir) is a broadspectrum oral antiviral originally developed by FUJIFILM Toyama Chemical Co, Ltd (FFTC) and approved in Japan as a treatment and stockpile countermeasure for pandemic influenza Favipiravir disrupts viral replication by acting directly on the viral RNA polymerase enzyme Favipiravir brings several advantages Extensive clinical Jul Dr Reddys acquires favipravir This is the second deal by Dr Reddys for a COVID19 treatment within days following the nonexclusive agreement with Gilead Sciences for marketing remdesivir Unclear at this stage whether there are plans to develop for influenza (currently available in Japan for this indication) 1113Favipiravir is a pyrazine carboxamide derivative (6fluoro3hydroxy2pyrazinecarboxamide) and a broadspectrum antiviral drug approved in Japan for the treatment of influenza4 Favipiravir is a prodrug that is ribosylated and phosphorylated intracellularly to form the active metabolite favipiravir ibofuranosyl5′triphos phate (T705RTP)4 T705RTP competes with purine




Glenmark Launches Fabiflu Favipiravir As The First Oral Medication To Treat Mild To Moderate Covid 19 In India Pharmashots




Favipiravir T 705 A Broad Spectrum Inhibitor Of Viral Rna Polymerase Abstract Europe Pmc
This is an open label, randomized, controlled, multicenter Phase 2 proofofconcept study of favipiravir in hospitalized subjects with COVID19 Subjects will be randomized within their study site and stratified by the severity of their disease to receive either favipiravir standard of care (SOC) or SOC alone The dose regimen will be 1800 mg favipiravir BID plus SOC or SOCFavipiravir tablets should be swallowed whole with water The tablet should not be opened, broken, or chewed For patients with COVID19, the usual dosage of favipiravir for adults is 1800 mg orally twice daily on day 1, followed by 800 mg orally twice daily for 14 days FIR in fake favipiravir racket Ahmedabad A complaint of attempt to culpable homicide was filed against Sudip Mukherjee, owner of Max Relief Healthcare, for his alleged involvement in a multi




Early Favipiravir Treatment Was Associated With Early Defervescence In Non Severe Covid 19 Patients Journal Of Infection And Chemotherapy



Fujifilm S Flu Drug Favipiravir Sent To 43 Nations For Covid 19 Trials
Favipiravir is an antiviral drug that has been licensed in Japan since 14 to treat influenza Favipiravir has the same effect as Remdesivir which aims to prevent the duplication of SARSCoV2 virus Many countries have approved clinical trials of this drug in the treatment of Covid19 patients Unclear effectsPrice A number of variables affect the total price of a medicineFavipiravir Please Note Medicines will be available to order by healthcare professionals only Therapeutic area (J) Antiinfectives for systemic use Strength and dosage form(s) If the strength or dosage form you require is not listed, please complete a sourcing request or contact our customer services team 0 mg Tablet;




High Doses Of Favipiravir Counter Sars Cov 2 In Hamsters Hydroxychloroquine Has No Significant Effect Study



Japanese Drug Favipiravir Proving Effective Against Covid 19 In As Little As 4 Days Daily Sabah
Favipiravir () is an oral antiviral drug used in the treatment of influenza virus in Japan Favipiravir was first discovered in a laboratory screening for potential antiviral properties against the influenza virus by Toyama Chemical Company, Ltd Favipiravir a prodrug, is converted intracellularly into its active form (favipiravir Favipiravir (T705, Favipira) Therapeutic uses, Clinical uses, Indications, Dosage, Side Effects, Warnings, Interactions, Precautions, Contraindications & Brand names Updated on 22nd April These materials are regularly updated based on new scientific findings & trials as the pandemic evolves Favipiravir, a pyrazine carboxamide derivative, is an antiviral medication Favipiravir is a pyrazine carboxamide derivative (6fluoro3hydroxy2pyrazinecarboxamide) and a broadspectrum antiviral drug approved in Japan for the treatment of influenza 4 Favipiravir is a prodrug that is ribosylated and phosphorylated intracellularly to form the active metabolite favipiravir ibofuranosyl5′triphosphate (T705RTP) 4 T705RTP




Favipiravir Quickens Recovery Show Trials Health Hindustan Times




Favipiravir Wikipedia
Favipiravir is a welltolerated drug with wellestablished safety profile Favipiravir demonstrates a lower proportion of Grade 1–4 adverse events (AEs),Gastrointestinal AEs, Serious AEs and an overall safety profile to comparators (mostly placebo) The most frequent adverse effects reported in clinical trials were increased serum uric acid level, diarrhoea, decrease of Avigan (Favipiravir) Antiviral Description Avigan (Favipiravir) () Tablet is a pyrazinecarboxamide derivative with activity against RNA virusesFavipiravir is converted to the ribofuranosyltriphosphate derivative by host enzymes and selectively inhibits the influenza viral RNAdependent RNA polymerase Avigan (Favipiravir) was approved for manufacture and sale Favipiravir is the first oral antiviral drug treatment option for mild to moderate COVID19 cases An informed consent form is provided with each pack of the medicine Favipiravir was primarily used to treat influenza The use of this drug was considered only when an outbreak of novel or reemerging influenza virus infection, which was not



Efficacy And Safety Of New And Emerging Drugs For Covid 19 Favipiravir And Dexamethasone Springerlink



Efficacy Of Favipiravir In Covid 19 Treatment A Multi Center Randomized Study Springerlink
Favipiravir (T705) is a synthetic prodrug, first discovered while assessing the antiviral activity of chemical agents active against the influenza virus in the chemical library of Toyoma chemicals A lead compound, A/PR/8/34, later designated as T1105, and its derivatives were found to have antiviral activities Favipiravir is originally approved in Japan to treat influenza It is now expected to effectively mitigate the severity of symptoms and shorten the duration of Covid19 Stanford Medicine clinical professor Aruna Subramanian said "We hope that this drug can help to reduce transmission within families, groups and schoolFavipiravir Favipiravir Favipiravir (T705) is a pyrazinecarboxamide derivative with activity against RNA viruses Favipiravir is converted to the ribofuranosyltriphosphate derivative (favipiravirribofuranosyl5'triphosphate) by host enzymes and selectively inhibits the influenza viral RNAdependent RNA polymerase




Cipla To Launch Favipiravir Under Ciplenza In August At 68 Rupees Per Pill




Favipiravir C5h4fn3o2 Pubchem
Favipiravir is an antiviral medicine It stops the SARSCoV2 virus from multiplying By doing so, it decreases viral load in the body, restricts the spread of infection and helps in speeding up the recovery process Common side effects of Favipiravir Description Favipiravir (T705) is a potent and selective RNAdependent RNA polymerase inhibitor, used to treat influenza virus infections Targets RNAdependent RNA polymerase 1 In vitro Favipiravir shows antiinfluenza virus activities with IC50 ranged from 0013 to 048 μg/ml for the influenza A viruses, from 0039 to 00 μg/ml for Favipiravir (Avigan) is a known antiviral for influenza It is indicated for novel influenza strains that cause more severe disease rather than seasonal influenza Favipiravir triphosphate is a purine nucleoside analog Favipiravir is



Favipiravir And The Battle Against Covid 19 Scientific Update Uk




Explainer What Do We Know Of Remdesivir Favipiravir Dexamethasone And Other Drugs Used For Covid
Favipiravir led to shorter latencies to relief for both pyrexia (difference 170 days, P Favipiravir possibly exerted no significant beneficial effect in the term of mortality in the general group of patients with mild to moderate Favipiravir, previously known as T705, is a prodrug of the purine nucleotide favipiravir ribofuranosyl5′triphosphate The active agent




Fujifilm Plans To Make Flu Drug Avigan Available For 2 Million People By March 21 The Japan Times




Favipiravir Very Effective Against Covid 19 Clinical Trials Show Cgtn
Favipiravir is an antiviral drug that has been licenced in Japan since 14 to treat influenza It works by inhibiting a viral enzyme called RNA polymerase, preventing viral replication within human cells This viral enzyme is common to several viruses, including SARSCoV2, which causes COVID19 Favipiravir adalah obat antivirus yang digunakan untuk mengatasi beberapa jenis virus influenza, seperti influenza A, yang menyebabkan flu burung dan flu babi, inluenza B, dan influenza C Saat ini, favipiravir juga sedang diteliti lebih lanjut Favipiravir (prodrug) is a purine base analog that is converted to active favipiravir ribofuranosyl5Btriphosphate (favipiravirRTP) by intracellular phosphoribosylation It is a selective and potent inhibitor of RNAdependent RNA polymerase (RdRp) of RNA viruses




Favipiravir Use In Covid 19 Analysis Of Suspected Adverse Drug Events Idr




Remdesiver Favipiravir Antiviral Drugs No Game Changers In Covid 19 Fight Say Medical Experts India News India Tv
Overall, favipiravir has shown promising results in clinical studies in China, Russia, and Japan, and more trials are underway in multiple countries, including USA, UK, and India Recently, treatment guidelines from many countries and some states from India have included favipiravir in the treatment protocol This review provides insights into the evidencebased evolving role of favipiravirHowever, there was no evidence that favipiravir monotherapy was effective 2 Preclinical data against influenza FPV was orally administered to mice BID at dosages of 50, 150, or 300 mg/kg/day for 5 days and oseltamivir was used as control FPV protected mice from lethal infection in a dosedependent manner FPV can be effective toUdwadia 21) Based on data from animal reproduction studies, use is contraindicated for use in patients who may become pregnant Pregnancy status should be evaluated prior to use Patients who may become pregnant should use effective contraception



Coronavirus Drug Launched For Moderate Covid 19 Cases The Hindu




Coronavirus Medicine Covid Drug News Glenmark Defends Price Of Its Covid 19 Drug Favipiravir Version
Thailand needs about 300,000 Favipiravir tablets each day, or about 9 million a month Earlier, GPO had imported an additional 55 million




The Mechanism Of Resistance To Favipiravir In Influenza Pnas




Favipiravir A New And Emerging Antiviral Option In Covid 19 Sciencedirect




Favipiravir And Remdesivir Appear To Be Relatively Unencumbered With Drug Drug Interactions Anesthesia Patient Safety Foundation



Early Treatment With Favipiravir Shows Faster Viral Clearance In Mild Covid 19 Cases Study Cities News The Indian Express




Favipiravir Led To 40 Faster Cure From Covid Glenmark




Fujifilm Tests Favipiravir As A Covid 19 Treatment



Avigan Influenza Antiviral To Enter Phase Iii Trials In Covid 19 Patients




Favipiravir To Be Investigated As A Possible Covid 19 Treatment For At Home Recovery In The Principle Trial Principle Trial




Favipiravir An Anti Influenza Drug Against Life Threatening Rna Virus Infections Sciencedirect




Favipiravir Permanently Registered In Russia For Covid 19 Treatment Society Culture Tass




Covid 19 Treatment Fabiflu By Glenmark Hcq Remdesivir And Other Drugs That Are Being Used To Treat Coronavirus Patients Health News Firstpost




Efficacy And Safety Of Favipiravir An Oral Rna Dependent Rna Polymerase Inhibitor In Mild To Moderate Covid 19 A Randomized Comparative Open Label Multicenter Phase 3 Clinical Trial International Journal Of Infectious Diseases




Clinical Efficacy And Safety Of Favipiravir In The Treatment Of Covid 19 Patients Journal Of Infection



Avifavir First Covid 19 Drug From Russia What You Need To Know Special Reports Gulf News




Glenmark Approved To Supply Favipiravir As Covid 19 Treatment In India




Favipiravir A New And Emerging Antiviral Option In Covid 19 Sciencedirect



Favipiravir And The Battle Against Covid 19 Scientific Update Uk




Covid 19 Pune Based Drug Firm To Export Favipiravir To 18 Countries Business Standard News




Favipiravir For Covid 19 In A Patient On Hemodialysis American Journal Of Kidney Diseases
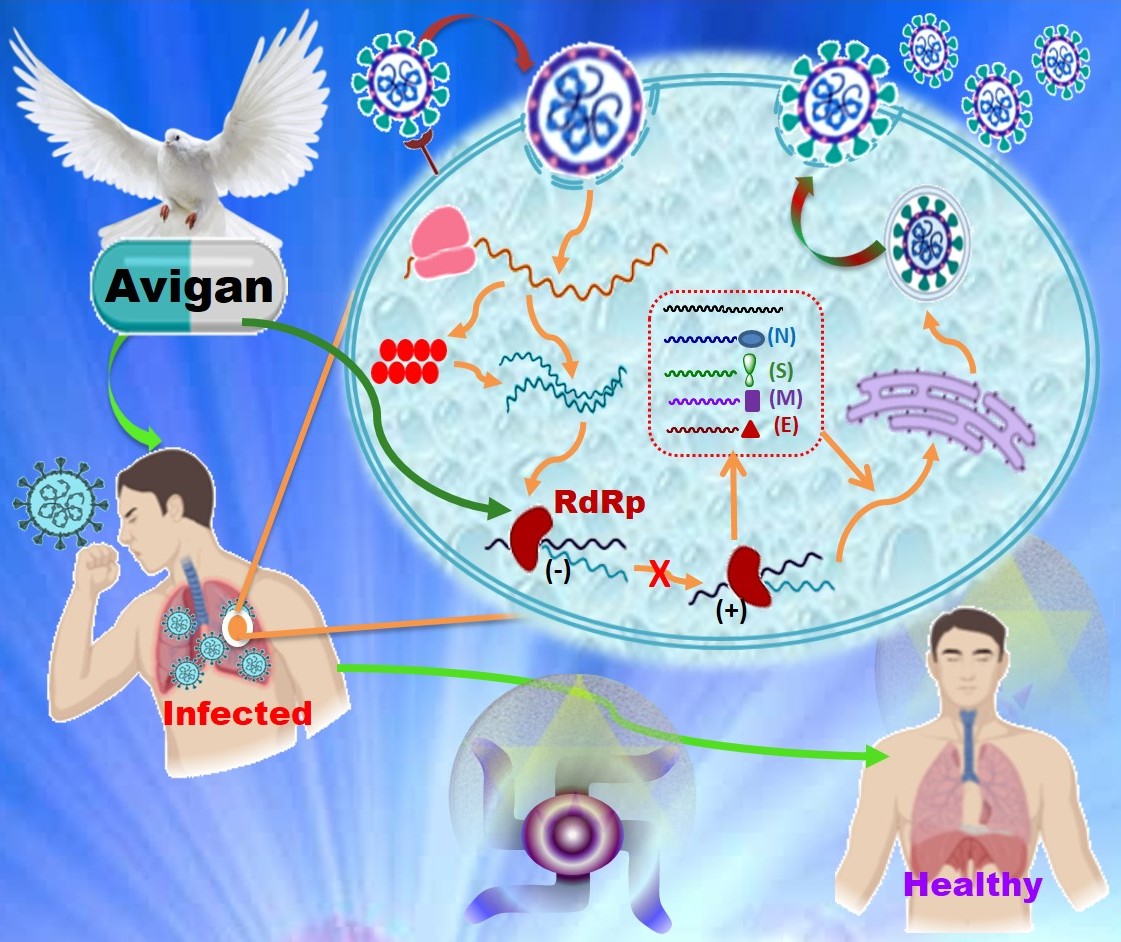



A Systematic Review Of The Efficacy And Safety Of Favipiravir Avigan For The Treatment Of Novel Covid 19 Infections Medical Research Archives




High Dose Favipiravir Countered Sars Cov 2 In Hamsters Hydroxychloroquine Ineffective Global Biodefense




A Case Of Covid 19 Pneumonia Successfully Treated With Favipiravir Avigan In Which Serum Sars Cov 2 Rna Detected By Lamp Method Was Clinically Useful Journal Of Infection And Chemotherapy




Favipiravir Antiviral Drug To Fight Coronavirus Databaaz Govindraj Ethiraj Omkar Herlekar Youtube
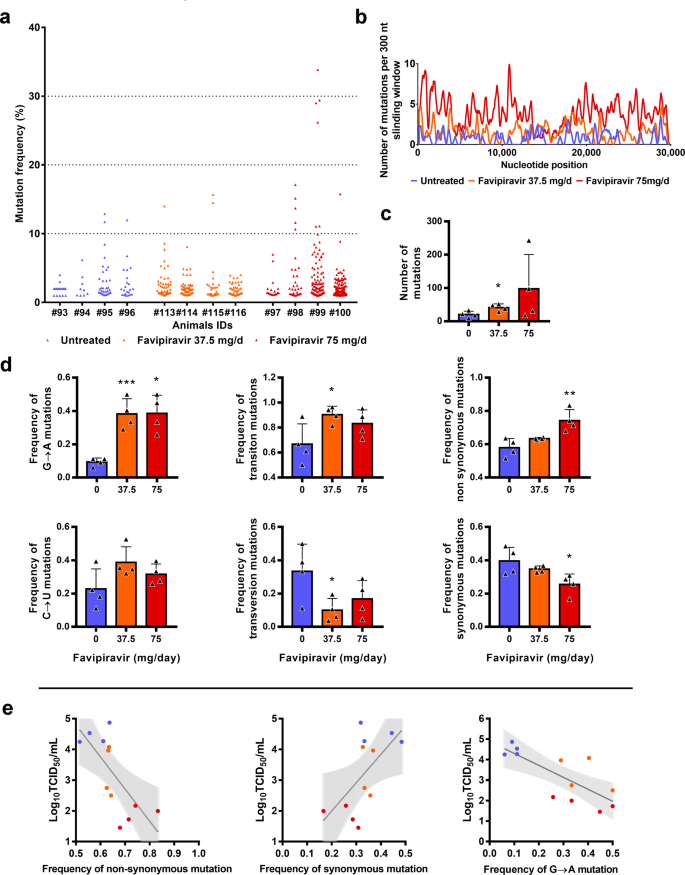



Favipiravir Antiviral Efficacy Against Sars Cov 2 In A Hamster Model Nature Communications
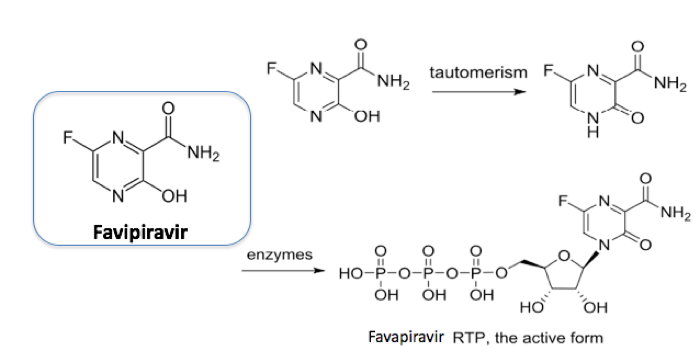



Favipiravir And The Battle Against Covid 19 Scientific Update Uk




News Favipiravir To Be Investigated As A Possible Covid 19 Treatment For At Home Recovery In The Principle Trial Nihr



Prices Of Remdesivir And Favipiravir Being Monitored In India Says Report
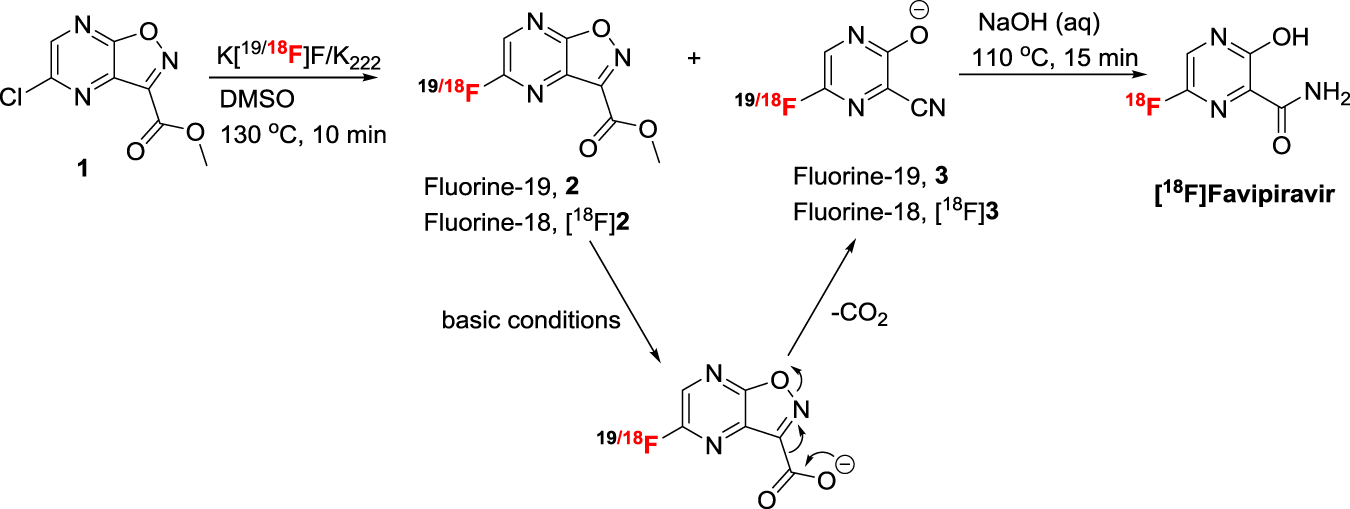



Synthesis Of 18 F Favipiravir And Biodistribution In C3h Hen Mice As Assessed By Positron Emission Tomography Scientific Reports




Pharma Giant To Start Favipiravir Clinical Trials For Covid 19 How Does It Work Times Of India




Glenmark Pharma Gets Nod For Favipiravir Cipla Hetero For Remdesivir Business Standard News




Efficacy And Safety Of Favipiravir An Oral Rna Dependent Rna Polymerase Inhibitor In Mild To Moderate Covid 19 A Randomized Comparative Open Label Multicenter Phase 3 Clinical Trial International Journal Of Infectious Diseases




Favipiravir An Anti Influenza Drug Against Life Threatening Rna Virus Infections Sciencedirect




Glenmark Reports Positive Data Of Favipiravir In Covid 19 Pms Study




Glenmark S Antiviral Drug Favipiravir Boosts Recovery Time Of Covid 19 Patients In Phase 3 Study Covid 19 Hospimedica Com




Covid 19 Treatment Favipiravir Drug Launched As Fabiflu By Glenmark Pharmaceuticals
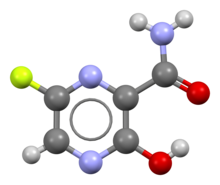



Favipiravir Wikipedia




Bmc Shells Out High Price For Favipiravir When State Government Buys Covid 19 Oral Drug For Rs 54 Per Tablet




Favipiravir Use In Covid 19 Analysis Of Suspected Adverse Drug Events Idr




Glenmark Starts Phase Iii Favipiravir Combination Trial For Covid 19




Medical Experts On Remdesiver Favipiravir For Covid Treatment No Game Changers




Influenza Drug Good Contender For At Home Treatment Against Covid 19 The Independent




Role Of Favipiravir In The Treatment Of Covid 19 International Journal Of Infectious Diseases




Drl To Launch Innovator Brand Of Favipiravir Is Silent On Pricing Business Standard News



Favipiravir Helps In Mild To Moderate Covid 19 Cases Reduces Treatment Time Study Latest News India Hindustan Times



Glenmark Releases Phase 3 Trial Data Of Favipiravir Says 40 Faster Clinical Cure Witnessed Coronavirus News India Tv




Favipiravir And The Need For Early Ambulatory Treatment Of Sars Cov 2 Infection Covid 19 Antimicrobial Agents And Chemotherapy



Favipiravir Latest News Videos And Favipiravir Photos Times Of India




Drug Firms Race To Launch Favipiravir As Antiviral Gains Traction In Mild Moderate Covid 19 Care




Is Favipiravir Good For Covid 19 Clinical Trial Says No Press Release Says Yes The Wire Science




Favipiravir And Remdesivir Appear To Be Relatively Unencumbered With Drug Drug Interactions Anesthesia Patient Safety Foundation
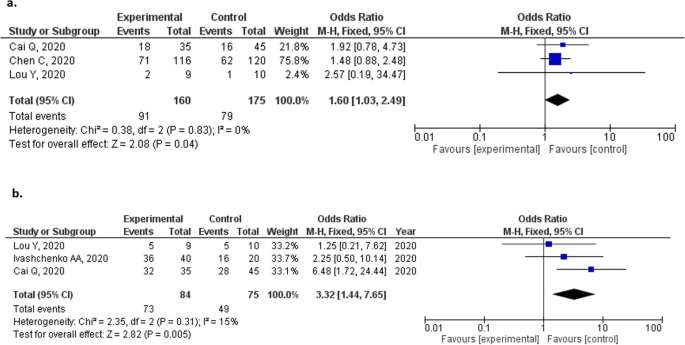



Favipiravir For The Treatment Of Patients With Covid 19 A Systematic Review And Meta Analysis Bmc Infectious Diseases Full Text




Avigan Antiviral Being Tested For Coronavirus Patients




Advanced Researches On The Inhibition Of Influenza Virus By Favipiravir And Baloxavir Sciencedirect



Favipiravir Has Limited Efficacy Data But Doctors Still Go For Glenmark S Drug To Cope With A Crisis The Economic Times



Japanese Flu Drug Appears Effective In Coronavirus Treatment In Chinese Clinical Trials Techcrunch



Favipiravir Does Not Attenuate The Progression Of Covid 19




Favipiravir Is Now The First Oral Covid 19 Treatment Approved In India All You Need To Know Health News Firstpost




Favipiravir For Coronavirus Treatment Check How Glenmark S Covid 19 Medicine Fabiflu Works The Financial Express



Covid 19 10 Hospitals Across India To Conduct Phase Iii Clinical Trial Of Favipiravir Drug Coronavirus Outbreak News The Indian Express




Stanford Researchers To Assess Favipiravir In Covid 19 Outpatients
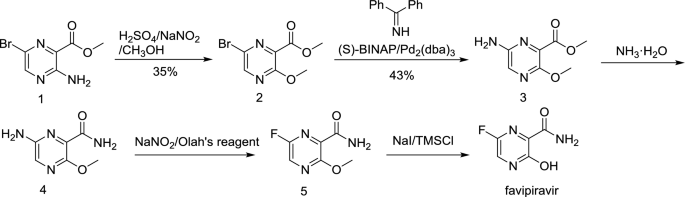



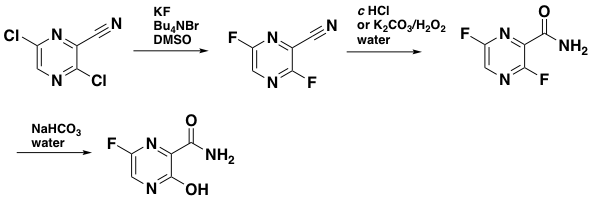
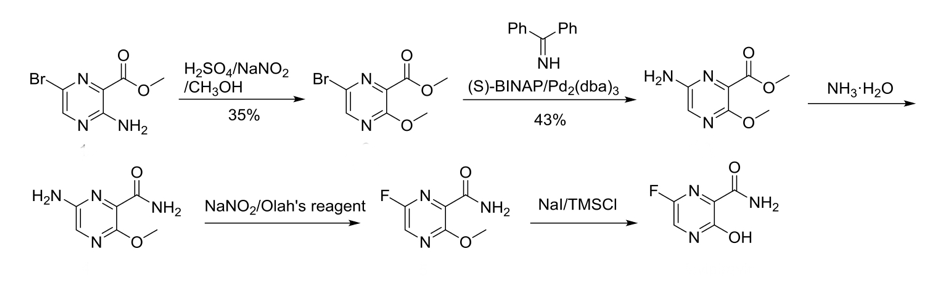
The Complete Synthesis Of Favipiravir From 2 Aminopyrazine Springerlink
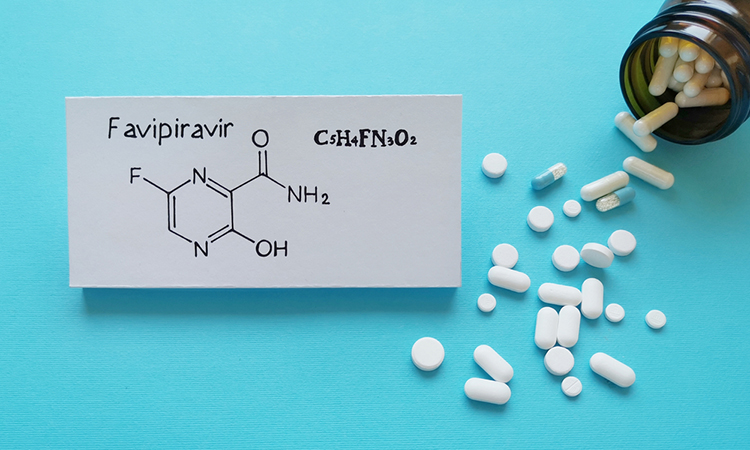



High Doses Of Favipiravir Combat Sars Cov 2 In Hamsters




The Mechanism Of Resistance To Favipiravir In Influenza Pnas
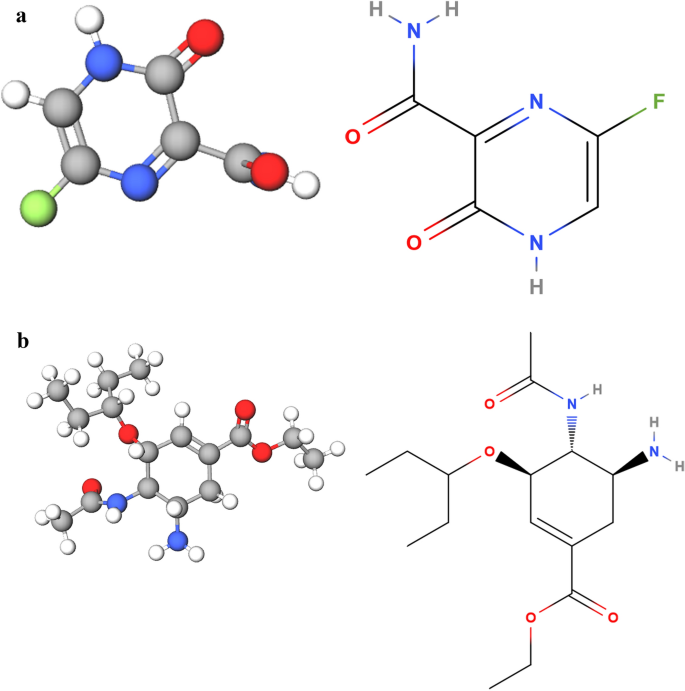



Favipiravir Use For Sars Cov 2 Infection Springerlink




Favipiravir Induced Fever In Coronavirus Disease 19 A Report Of Two Cases International Journal Of Infectious Diseases




Glenmark S Favipiravir Shows Improved Time To Cure In Covid 19 Trial




Japan Is Racing To Test Favipiravir A Drug To Treat Covid 19 Wired
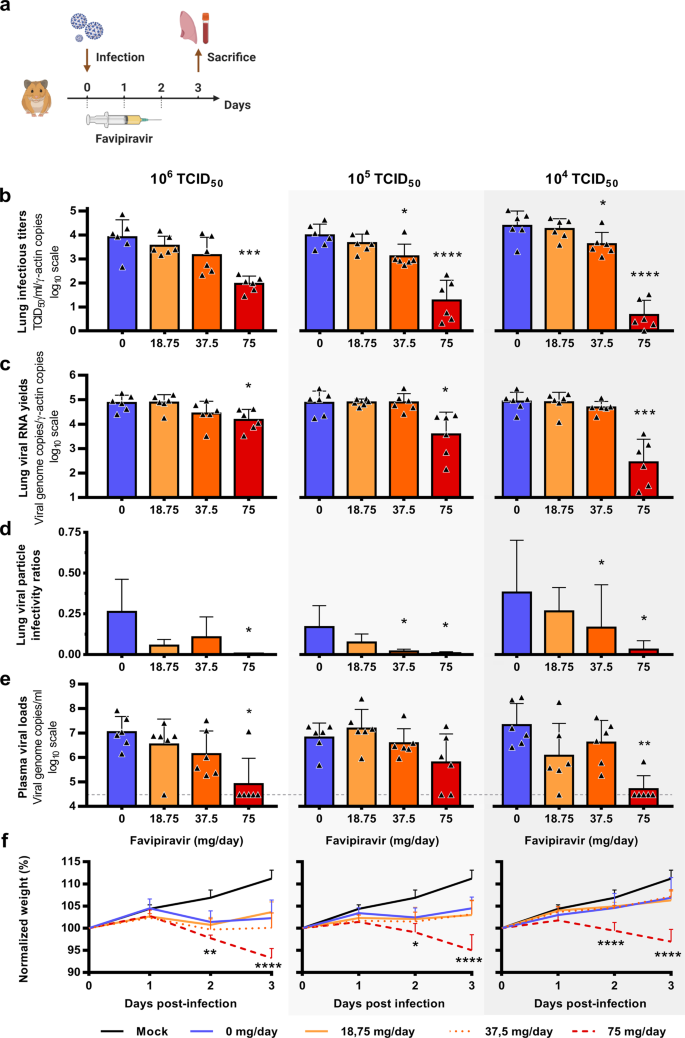



Favipiravir Antiviral Efficacy Against Sars Cov 2 In A Hamster Model Nature Communications




Favipiravir Sales Skyrocket In India Targeting Mild Covid 19 Cases




Role Of Favipiravir In The Treatment Of Covid 19 International Journal Of Infectious Diseases



Coronavirus Dr Reddy S Seeks Approval From Health Canada For Favipiravir The Hindu




Phase 2a Open Label Dose Escalating Multi Center Pharmacokinetic Study Of Favipiravir T 705 In Combination With Oseltamivir In Patients With Severe Influenza Ebiomedicine




Hetero Launches Generic Favipiravir To Treat Covid 19 In India




Zhejiang Hisun Shows Positive Effect Of Favipiravir On Covid 19
.jpg)



New Favipiravir Derivative Is A Potent Sars Cov 2 Inhibitor



Favipiravir Shows Good Clinical Efficacy In Treating Covid 19 Official Xinhua English News Cn


コメント
コメントを投稿